
Principles
Multiphoton-Lifetime Tomography of Human Skin
Multiphoton tomography of human skin exploits the fact that two-photon excitation in combination with non-descanned detection delivers optically sectioned images of tissue layers as deep as 100 µm. Three-dimensional tissue structures can be reconstructed from the data at sub-cellular resolution. The technique goes back to the work of Gratton, König, Masters, So and Tromberg, who showed that in-vivo two-photon imaging of cells and, especially, human skin is possible without impairing the viability of the tissue. By combining sub-µm resolution and optical sectioning with FLIM, morphological information is combined with biochemical and metabolic information. An impressive number of papers is related to this technique, see list of publications under ‘References’.
An example of multiphoton FLIM of skin in different depth is shown in the figure below. The upper row of images shows the stratum corneum, i.e. the uppermost layer of the skin. The lower row shows the stratum spinosum, about 50 µm below the surface.

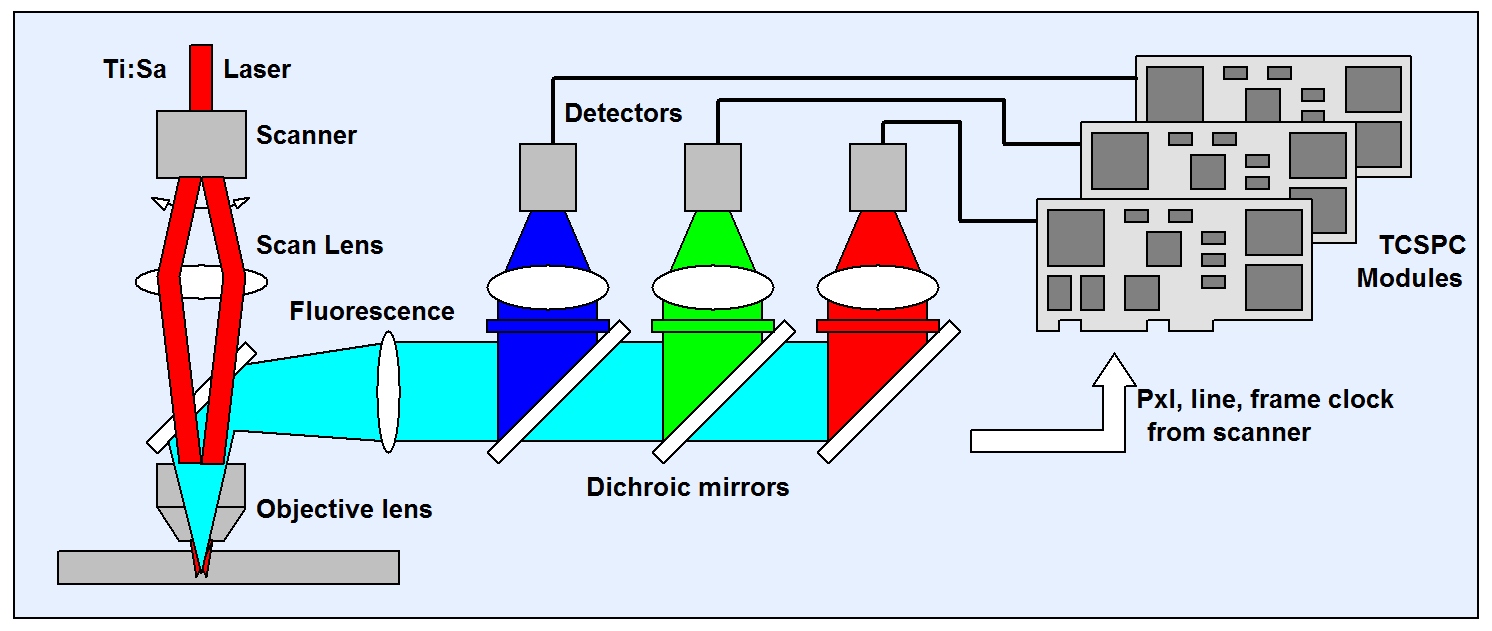
Instruments for clinical use have been developed by Jenlab, Germany. The instruments simultaneously detect in several wavelength intervals. The optical principle is that of a multiphoton microscope with non-descanned detection. The fluorescence light collected by the microscope lens is split off by a dichroic mirror, split into several spectral components by dichroic mirrors, and detected by several detectors. The FLIM data are recorded by one or several bh TCSPC FLIM modules. Earlier Instruments used a single bh SPC‑830 TCSPC FLIM module with a router. Later, the instruments have been upgraded with more recent SPC‑153 or SPC-180NX (three-channel) parallel TCSPC systems to record at higher count rates (and thus achieve shorter acquisition times). The general principle is shown in the figure below.
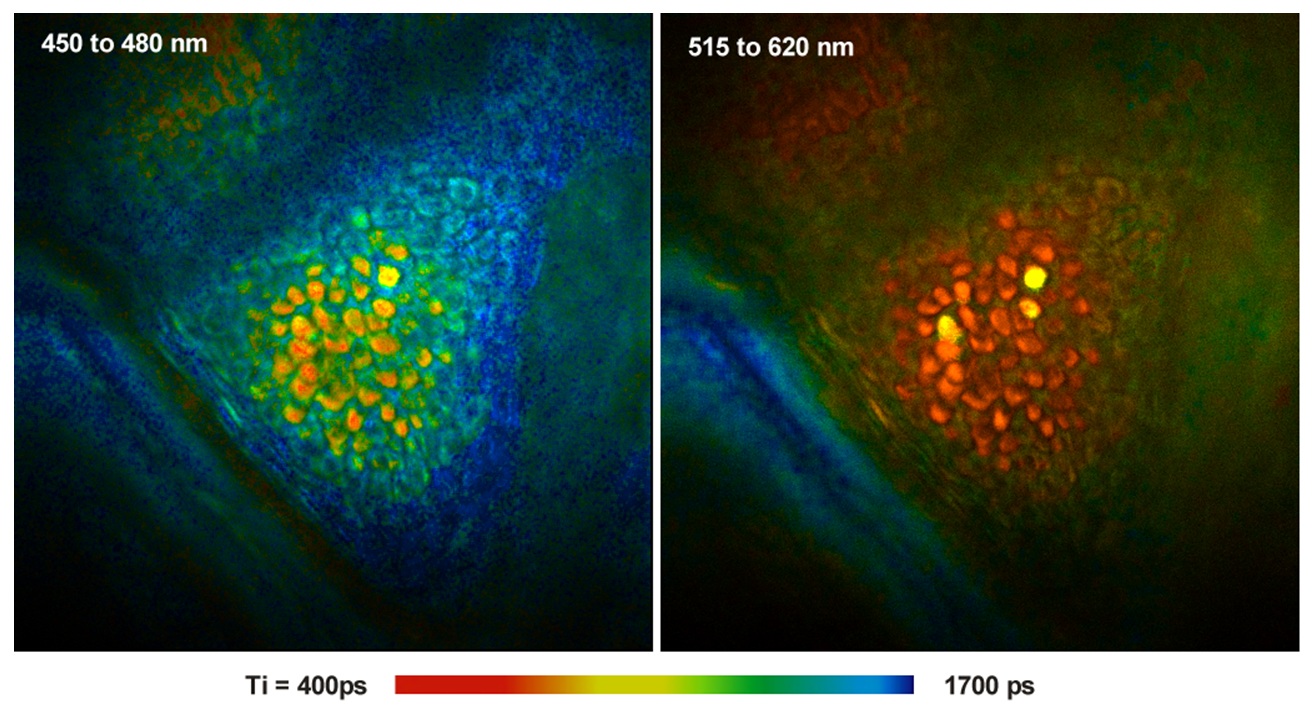
An image of the stratum corneum of a human volunteer recorded with this system is shown in the next figure. The excitation wavelength was 750 nm. The left image was recorded in the emission wavelength range of NAD(P)H, the right one in the emission range of melanin.
For details please see
- Becker, The bh TCSPC Handbook, chapter ‘Multiphoton Tomography of Human Skin’.
- Sanchez, M. Pastore, I. Haridass, K. König, W. Becker, M. S. Roberts, Fluorescence Lifetime Imaging of the Skin. In: W. Becker (ed.) Advanced time-correlated single photon counting applications. Springer, Berlin, Heidelberg, New York (2015)
- König, Clinical multiphoton tomography. J. Biophoton. 1, 13–23 (2008)
- Weinigel, H.-G. Breuning, A. Uchugonova, K. König, Multipurpose nonlinear optical imaging system for in vivo and ex vivo multimodal histology. Journal of Medical Imaging 2, 016003-1 to -10 (2015)
Please see also list of related publications under ‘References’.
References
References for In Vivo Imaging of Skin
- Dancik, A. Favre, C. J. Loy, A.V. Zvyagin, M.S. Roberts, Use of multiphoton tomography and fluorescence lifetime imaging to investigate skin pigmantation in vivo. J. Biomed. Opt. 18(2) 026022-1 to -13 (2013)
- Dimitrow, I. Riemann, A. Ehlers, M. J. Koehler, J. Norgauer, P. Elsner, K. König, M. Kaatz, Spectral fluorescence lifetime detection and selective melanin imaging by multiphoton laser tomography for melanoma diagnosis. Experimental Dermatology 18, 509-515 (2009)
- Dimitrow, M. Ziemer, M. J. Koehler, J. Norgauer, K. König, P. Elsner, M. Kaatz, Sensitivity and Specificity of Multiphoton Laser Tomography for In Vivo and Ex Vivo Diagnosis of Malignant Melanoma. J. Invest. Dermatol. 12(7) 1752-1758 (2009)
- Geusens, M. Van Gele, S. Braat, S.C. De Smedt, M.C.A. Stuart, T.W. Prow, W. Sanchez, M. Roberts, N.N. Sanders, J. Lambert, Flexible nanosomes (SECosomes) enable efficient siRNA delivery in cultured primary skin cells and in the viable epidermis of ex vivo human skin. Adv. Funct. Mater. 20, 4077-4090 (2010)
- D. Giorgi, D. Massi, S. Sestini, R. Ricci, F.S. Pavone, T. Lotti, Combined non-linear laser imaging (two-photon excitation fluorescence microscopy, fluorescence lifetime imaging microscopy, multispectral microscopy) in cutaneous tumors: First results. JEADV 23, 314-316 (2009)
- M. Holmes, J. Lim, H. Studier M.l S. Roberts, Varying the morphology of silver nanoparticles results in differential toxicity against microorganisms, HaCaT keratinocytes and affects skin deposition. Nanotoxixology 10, 1503-1514 (2016)
- J. Koehler, K. König, P. Elsner, R. Bückle, M. Kaatz, In vivo assessment of human skin aging by multiphoton laser scanning tomography, Opt. Lett. 31, 2879-2881 (2006)
- König, I. Riemann, High-resolution multiphoton tomography of human skin with subcellular spatial resolution and picosecond time resolution, J. Biom. Opt. 8, 432-439 (2003)
- König, Clinical multiphoton tomography. J. Biophoton. 1, 13–23 (2008)
- König, M. Weinigel, D. Hoppert, R. Bückle, H. Schubert, M.J. Köhler, M. Kaatz, P. Elsner, Multiphoton tissue imaging using high-NA microendoscopes and flexible scan heads for clinical studies and small animal research. J. Biophoton. 1, 506-513 (2008)
- Koenig, A. Uchugonova, Multiphoton Fluorescence Lifetime Imaging at the Dawn of Clinical Application. In: A. Periasamy, R.M. Clegg, eds., FLIM Microscopy in Biology and Medicine. CRC Press 2009
- R. Leite-Silva, D. C. Liu1, W. Y. Sanchez, H. Studier, Y. H. Mohammed, A. Holmes, W. Becker, J. E. Grice, H. A.E. Benson, M. S. Roberts, Effect of flexing and massage on in vivo human skin penetration and toxicity of zinc oxide nanoparticles. Nanomedicine (Lond.) 11(10), 1193–1205 (2016)
- R. Leite-Silva, W.Y. Sanchez, H. Studier, D.C. Liu, Y.H. Mohammed, A.M. Holmes, E.M. Ryan, I.N. Haridass, N.C. Chandrasekaran, W. Becker, J.E. Grice, H.A.E. Benson, M.S. Roberts, Human skin penetration and local effects of topical nano zinc oxide after occlusion and barrier impairment. European Journal of Pharmaceutics and Biopharmaceutics 104, 140-147 (2016)
- L. Lin, J. E. Grice, M.K. Butler, A.V. Zvyagin, W. Becker, T. A. Robertson, H.P. Soyer, M. S. Roberts, T.W. Prow, Time-correlated single photon counting for simultaneous monitoring of zinc oxide nanoparticles and NAD(P)H in intact and barrier-disrupted volunteer skin. Pharm Res. DOI 10.1007/s11095-011-0515-5 (2011)
- N. Pastore, H. Studier, C.S. Bonder, M.S. Roberts, Non-invasive metabolic imaging of melanoma progression. Exp. Dermatol. 26, 607–614 (2017)
- Patalay, C. Talbot, Y. Alexandrov, I. Munro, M. A. A. Neil, K. König, P. M. W. French, A. Chu, G. W. Stamp, C. Dunsby, Quantification of cellular autofluorescence of human skin using multiphoton tomography and fluorescence lifetime imaging in two spectral detection channels. Opt. Expr. 2, 3295-3308 (2011)
- Tarl W. Prow, Jeffrey E. Grice, Lynlee L. Lin, Rokhaya Faye, Margaret Butler, Wolfgang Becker, Elisabeth M.T. Wurm, Corinne Yoong, Thomas A. Robertson, H. Peter Soyer, Michael S. Roberts, Nanoparticles and microparticles for skin drug delivery. Advanced Drug Delivery Reviews 63, 470–491 (2011)
- Pires, M. S. Nogueira, S. Pratavieira, L. T. Moriyama, C. Kurachi, Time-resolved fluorescence lifetime for cutaneous melanoma detection. Biomed. Opt. Expr. 5, 2014, 3080-3089
- S. Roberts, M.J. Roberts, T.A. Robertson, W. Sanchez, C. Thorling, Y. Zou, X. Zhao, W. Becker, A. V. Zvyagin, In vitro and in vivo imaging of xenobiotic transport in human skin and in the rat liver. J. Biophoton. 1, 478-493 (2008)
- Roberts, M. S., Dancik, Y., Prow, T.W., Thorling, C.A., Li, L., Grice, J.E., Robertson, T.A., König, K., Becker,W. Non-invasive imaging of skin physiology and percutaneous penetration using fluorescence spectral and lifetime imaging with multiphoton and confocal microscopy. European Journal of Pharmaceutics and Biopharmaceutics 77, 469-488 (2011)
- Y. Sanchez, C. Obispo, E. Ryan, J. E. Grice, M.S. Roberts, Changes in the redox state and endogenous fluorescence of in vivo human skin due to intrinsic and phot-induced aging, measured by multiphoton tomography with fluorescence lifetime imaging. J. Biomed. Opt. 18(6), 061217-1 to -12 (2013)
- Y. Sanchez, M. Pastore, I. Haridass, K. König, W. Becker, M. S. Roberts, Fluorescence Lifetime Imaging of the Skin. In: W. Becker (ed.) Advanced time-correlated single photon counting applications. Springer, Berlin, Heidelberg, New York (2015)
- A. Thorling, A. Holmes, H. Studier, D. Liu, X. Liang, M. S. Roberts, Multiphoton and Fluorescence Lifetime Imaging Microscopy in Studying Nanoparticle Pharmacokinetics in Skin and Liver. In: Micro and Nanotechnologies for Biotechnology, http://dx.doi.org/10.5772/63452
- Weinigel, H.-G. Breuning, A. Uchugonova, K. König, Multipurpose nonlinear optical imaging system for in vivo and ex vivo multimodal histology. Journal of Medical Imaging 2, 016003-1 to -10 (2015)
- Zhu, C.-S. Choe, S. Ahlberg, M. C. Meinke, U. Alexiev, J. Lademann, M. E. Darvina, Penetration of silver nanoparticles into porcine skin ex vivo using fluorescence lifetime imaging microscopy, Raman microscopy, and surface-enhanced Raman scattering microscopy. J. Biomed. Opt. 20(5), 051006-1 to -8 (2015)
